The Jasmonate Code
What is the idea behind the project?
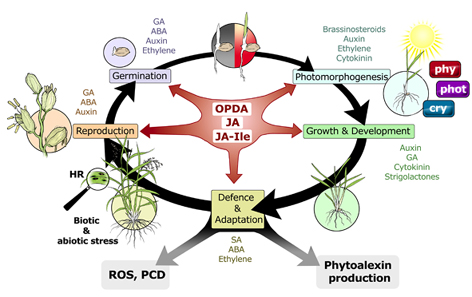
Jasmonic acid is a kind of plant adrenalin. How can such a general signal mediate so many different responses? The Jasmonate pathway is not linear, but highly branched with respect to both, synthesis as signalling. We reckon that this branching might be the base for specificity. We also think that the "meaning" of the signal not only arises from its molecular nature, but also from its spatio-temporal distribution (signature model). We address this in rice, the most important staple crop on this planet, but also in grapes as crop relevant for Europe. The role of jasmonate signatures for the resistance against soil salinity or drought are of high practical relevance.
How did this idea originate?
More than 25 years ago, we discovered a rice mutant (hebiba - "snake leaf") that mixes up light and dark. Surprisingly, this mutant turned out to be deficient in jasmonate (). After many years of genetic mapping and complementation, we could show that in this mutant the gene Allene-Oxid-Cyclase (AOC) was deleted (Riemann et al., 2013). In addition to the hebiba mutant, we were able to isolate additional mutants in synthesis or processing of jasmonates. With the help of these mutants we were able to decipher numerous stress responses of rice.
What is our vision?
Different stress contexts require different adaptive responses. How can we get specificity on the base of jasmonate signalling? We want to test whether temporal and spatial patterns (signal signatures) are relevant for specificity. In fact, we were able to show that temporal shifts produce different response qualities. In analogy with human language, where the arrangement of words can convey different meaning, we want to decipher the rules of "plant stress grammar". We are therefore analysing the rise and fall of signals over time, and we try to shift temporal patterns by integration of gene switches. We were already able to show that the precursor OPDA launches a branch in the signal that activates programmed cell death (a plant version of apoptosis). When we succeed to control this branch, we might be able to "persuade" rice to produce yield under stress rather than to activate necrosis.
Publications
- Svyatyna K., Riemann M. (2012) Light-dependent regulation of the jasmonate pathway. Protoplasma 249 Suppl 2:S137-145
- 46.Riemann M, Müller A, Korte A, Furuya M, Weiler EW, Nick P (2003) The rice mutant hebiba lacks jasmonate induction. Plant Physiol 133, 1820-1830 - pdf - kurze Erklärung der Bedeutung dieser Arbeit finden Sie
- 95. Riemann M, Haga K, Shimizu T, Okada K, Ando S, Mochizuki S, Nishizawa Y, Yamanouchi U, Nick P, Yano M, Minami E, Takano M, Yamane H, Iino M (2013) Isolation of rice ALLENE OXIDE CYCLASE mutants and the function of jasmonate for defence against Magnaporthe oryzae. Plant J 74, 226–238 - pdf
- 101. Svyatyna K, Jikumaru Y, Brendel R, Reichelt M, Mithöfer A, Takano M, Kamiya Y, Nick P, Riemann M (2014) Light induces jasmonate-isoleucine conjugation via OsJAR1-dependent and -independent pathways in rice. Plant Cell Environment 37, 827-839 - pdf
- 106. Brendel R, Svyatyna K, Jikumaru Y, Reichelt M, Mithöfer A, Takano M, Kamiya Y, Nick P, Riemann M (2014) Effects of light and wounding on jasmonates in rice phyAphyC mutants. Plants 3, 143-159 - pdf
- [35] Ismail A, Takeda S, Nick P (2014) Life and death under salt stress: same players, different timing? J Exp Bot 65, 2963-2979 - pdf
- 113. Hazman M, Hause B, Eiche E, Nick P, Riemann M (2015) Increased tolerance to salt stress in OPDA-deficient rice ALLENE OXIDE CYCLASE mutants is linked to an increased ROS-scavenging activity. J Exp Bot 66, 3339-3352 - pdf
- 117. Ismail A, Seo M, Takebayashi Y, Kamiya Y, Nick P (2015) A Balanced JA/ABA status might correlate with adaptation to osmotic stress in Vitis cells. J Plant Physiology 185, 57-64 - pdf
- [39] Riemann M, Dhakarey R, Hazman M, Miro B, Kohli A, Nick P (2015) Exploring jasmonates in the hormonal network of drought and salinity responses, Frontiers Plant Sci 6, doi: 10.3389/fpls.2015.01077 - pdf
- 123. Hazman M, Hause B, Eiche B, Riemann M, Nick P (2016) Different forms of osmotic stress evoke qualitatively different responses in rice. J Plant Physiol 202, 45-56 - pdf
- 129. Chang X, Seo M, Takebayashi Y, Kamiya Y, Riemann M, Nick P (2017) Jasmonates are induced by the PAMP flg22 but not the cell death-inducing elicitor Harpin in Vitis rupestris. Protoplasma 254, 271-283 - pdf