Achillesferse eines Weinpathogens
What is the topic?
Downy Mildew represents the most serious challenge in viticulture in Central Europe. Around 80% of fungicides in Germany are needed for this pathogen, yield losses range depending on the season around 1/3. Due the intensive fungicide use fungicide-resistant strains have evolved already. Even the use of newly bred cultivars that are resistant against Downy Mildew will not be a sustainable solution – since around five years pathogen strains have been discovered that can break the resistance of such grape varieties. Our approach is to investigate the cell biology of this pathogen to develop novel strategies for plant protection.
Project
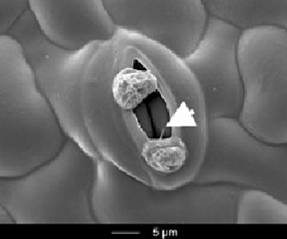
Most of the time Downy Mildew lives inside the leaf - well protected from fungicides. There is, however, a short phase, where it has to be at the surface of the leaf and we think that this phase provides the sensitive point, where we can catch it: From the sporangia, several flagellate zoospores hatch and have to find their path to the nearest Stoma on the surface of rain-wettened leaves. This phase in the life cycle is very short (less than 15 min!) and basically recapitulates the evolutionary history of this pathogen – Downy Mildew is not a fungus, but as a so called Oomycete a descendant of algal ancestors. The flagellate zoospore has to swim and therefore does not have a cell wall. During its path to the stoma, it will therefore constantly be challenged by influx of water. Similar to other freshwater protists, Plasmopara is endowed with a pulsating vacuole that will extrude the incoming water. If we would be able to block the function of this "cellular kidney", the spore should burst and the infection cycle would be interrupted...
Current state
We have started to investigate the pulsating vacuole with respect to structure and function. By fluorescent membrane trackers we can visualise this Organelle by spinning-disc confocal microscopy. The zoospores can be induced to hatch in vitro from sporangia. By specific Inhibitors, we were able to induce bursting and this can be quantified, such that dose-Response studies are possible. In addition to blockers of endocytosis(Wortmannin), and the actin cytoskeleton (Latrunculin B, Phalloidin), also Al- or Cu-ions were effective. Most recently, we could show that an extract from Glycyrrhiza uralensis, a plant used in the Traditional Chinese Medicine is effective. In cell cultures from grapevine, this extract also induces a strong apoplastic alkalinisation, which is an early readout for activated plant immunity.
In the focus
The effect of the Glycyrrhiza extract should be followed further. The major compound, glycyrrhizin, should be tested as well. In addition to its effect on zoospores (burst assay), the potential immunostimulating activity of this compound should be tested in cell cultures. For both effects detailed dose-response studies should be generated (if possible, with glycyrrhizin - when glycyrrhizin is not the responsible compound, we have to use the extract). In the second part, it will be asked, what the molecular mechanism might be. One working hypothesis is that both effects (bursting and immunostimulation) are linked with the induction of reactive oxygen species (ROS). ROS, on the one hand act as bundlers of actin (which might explain the block of the pulsating vacuole), on the other, they are important signals in plant defence. To address this point, we might test the effect of artificial ROS (hydrogen peroxide or Fenton reagent) or the effect of blocking ROS Formation (in plants possible using the inhibitor DPI).
Publikationen
70. Jürges G, Kassemeyer HH, Dürrenberger M, Düggelin M, Nick P (2009) The mode of interaction between Vitis and Plasmopara viticola Berk. & Curt. Ex de Bary depends on the host species. Plant Biol 11, 886-898 – pdf