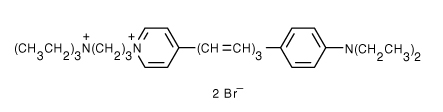
Name:
(N-(3-triethylammoniumpropyl)-4-(6-(4-(diethylamino)phenyl)hexatrienyl)pyridinium dibromide). Synonyms: FM4-64, Synapto-Red. FM stands for Fei Mao, the researcher at Molecular Probes, who developed this styryl-based dye.
Target:
- outer sheet of the plasma membrane, fluorescence develops only in lipophilic environment
- therefore little background, staining of plasma membrane and later endocytotic vesicles
- vital dye, in case that fixation is needed, there exist also variants at Molecular Probes
Chemical properties:
- MW 608
- purchasable by Molecular Probes (T13320)
- stock solution: 20 mM in DMSO, keep at -20°C, add sodium azide up to 15 mM to prolong lifetime. Others propose 1 mg/ml in water. Lifetime is around 6 months.
- in water, the stock solution is dark purple
- working concentrations in plant cells 2-10 µM depending on the application time
Application:
- FM4-64 is not fluorescent in aequous environment, fluorescence develops in a lipophilic environment.
- Therefore, background fluorescence is low, now washing needed.
- The dye is added to the cell and observation starts immediately, upon insertion into the outer sheet of the cell membrane, the dye is fluorescent.
- with prolonged observation, endocytotic vesicles become visible, therefore FM4-64 is often used as endocytotic tracer
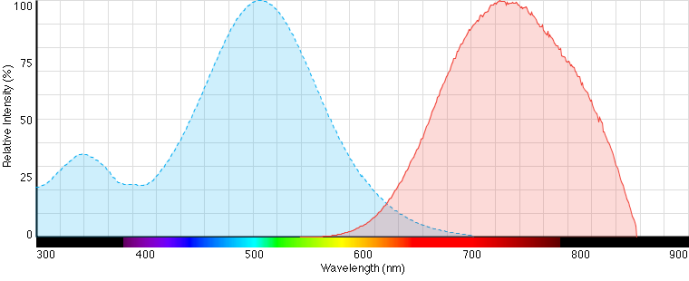
- excitation at 520 nm, fluorescence at 700 nm (a rhodamine set-up can be used)
- note: in case that a FITC set-up is required, a similar dye FM1-43 (SynaptoGreen) can be used instead
Caveat:
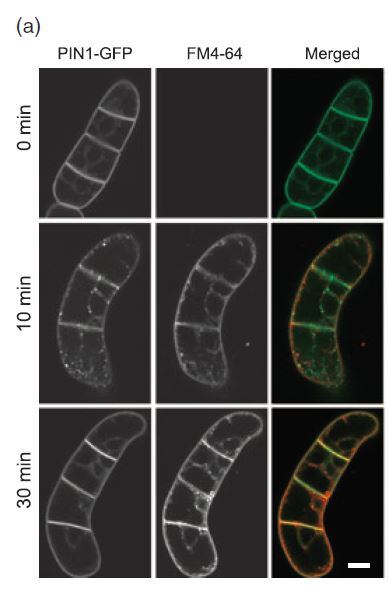
- like any fluorescent dye binding to a target, one has to be careful about side effects. This has been investigated in a good paper by Jelinková et al. (2010), application of FM4-64 has been found to induce a transient (peak around 10 min) internalisation of plasma membrane proteins (including PIN1). Later, when FM4-64 is seen to internalise strongly as endocytotic vesicles (at around 30 min), these plasma membrane proteins have returned to the membrane. The vesicles labelled by FM4-64 and those linked to PIN1 are different.
- the spectrum of the FM dyes is very broad. This means that in dual labelling optical bleed-through is an issue (i.e. the signal from the FM dye can also be seen in the other filter set). To account for this, negative controls with the FM dye alone are important to see the bleedthrough of this background.
References:
- Gaffield MA, Betz WJ (2006) Imaging synaptic vesicle exocytosis and endocytosis with FM dyes. Nature Protocols 1, 2916-2921 - pdf (gives a good overview about the chemistry and general applications, also in animal cells, the senior author developed this type of dyes together with Fei Mao at Molecular Probes
- Jelínková A, Malínská K, Simon S, Kleine-Vehn J, Parezova M, Pejchar P, Kubes M, Martinec J, Friml J, Zazímalová E, Petrásek J (2010) Probing plant membranes with FM dyes: tracking, dragging or blocking? Plant J 61, 883-892 - pdf (describes the transient internalisation of membrane proteins by FM dyes in different cell types. The results on inhibitor treatments - Latrunculin B, Wortmannin, Brefeldin A, Oryzalin - should be seen with certain scepticism, because important controls are lacking)
Publications from our group using this dye
73. Eggenberger K, Schröder T, Birtalan E, Bräse S, Nick P (2009) Passage of Trojan Peptoides into Plant Cells. ChemBioChem 10, 2504-2512 - pdf
98. Liu Q, Qiao F, Ismail A, Chang X, Nick P (2013) The Plant Cytoskeleton Controls Regulatory Volume Increase. BBA Membranes 1828, 2111–2120 - pdf